Understanding Fatty Acids

Every day we are confronted with mountains of information about our nutrition, and only very few people know what it all means! I do not say this in any derogative way – everyone has their area of expertise and interests! But I think following scientific advances and in this case drawing conclusions on how to eat healthily sometimes just gets too much – how is anyone supposed to remember all of it😊
I always thought I had a good attitude toward food – I ate regularly, I ate a little bit of everything, I didn’t have an overly dominant sweet tooth and coming from a wine-loving country, I also enjoyed drinking wine. But not until recently, where through a few converging circumstances I put on a large amount of weight, did I realize that I had to make a few changes in my diet as well as my lifestyle (also I hit my mid 30s..who would have thought that my mother was right when she said “One day you won’t be able to eat everything you like!!”).
Before I mostly I enjoyed eating quite rich food, which I traded to a more lighter and fresh diet! But what makes the difference? And how do the fats in our diet influence our health?
In all seriousness: in an era of a new-found consciousness about health and nutrition, I think it is hard to make an informed decision about what may be healthy for you or not, based on the information provided. And that is just considering the solid scientific facts – don’t get me started on the pseudo-science and scientific untruths that travel this very welcoming place called “the internet”!
I get very upset when I see people exploited and lied to for the sake of profit and other obscure reasons that make no sense. “Buy this or buy that and we guarantee you will shot wings out of your backside and achieve world peace.” ….
I consider it my duty to do my part: and that is why it is so important to me to use my 15 years of scientific studies and experience. I want to share some of the knowledge I gained over the years with you, at least in my little area of expertise – which is fat or more precisely lipid metabolism. We hear so many things every day: fat is good, fat is bad, fat is evil – but how do you know what it all means? I didn’t! At least not until I got into my biochemical studies.
This article will mostly focus on a very common word you hear these days: fatty acids. I bet you have heard at least one of the following statements in the last few months:
- Saturated fatty acids are bad!
- Unsaturated fatty acids are good
- Trans fatty acids are bad.
- You need Omega-3 fatty acids, because they are good for your brain!
Sound familiar?
I welcome you, dear reader, to take this short journey with me, so that at the end you will know that little bit more about what you give to your body!
This article is the introductory article to my eBook, if you want to find out more download it here!
Fact: Your body needs energy
It’s actually quite easy: we need energy, and our body gets this energy through our food. The human body can use different sources to gain energy – the most important ones being carbohydrates and fats from our diet. Carbohydrates can be eaten in form of potatoes, rice, pasta, bread but also sugar (refined or contained in fruits for example). Fats can be contained in many foods like meats, fish or certain vegetables or we can eat them in the form of butter or oils.
However, your body doesn’t use these nutrients directly to fuel your bodily functions, it transforms them into a universal “energy currency” called ATP, adenosine triphosphate. The energy your body gains through your nutrition is used to produce ATP. And if your body requires energy, ATP is used up (or broken down).
Imagine it like having a ball with 3 beads on a chain attached to that ball. If you require energy (for example to move your muscles or to think), one or more of these beads are separated from the rest, and the energy that is stored within the bond that links the beads is released.
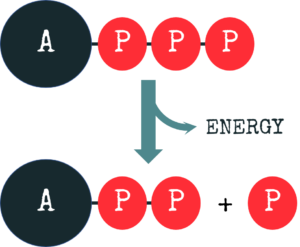
Let’s take a look at an example: from 1 molecule of glucose, a sugar contained in most of our foods, you can make 32 molecules of ATP. On the other hand, one fatty acid (let’s say the most common fatty acid palmitic acid) can yield up to 106 molecules of ATP when it is completely broken down (or oxidized). You may say sugar is the quicker source of energy, but fatty acids have a considerably higher yield!
The body will first use its available sugars or carbs for ATP production, then it will turn to fatty acids, and finally it can also use amino acids (which will not be addressed here). A practical example may describe your body’s reaction to exercise:
- If you want to increase your muscle mass, you might fill up on carbs (sugars) before your training – this way your body uses the sugars for ATP production, and your muscle mass will increase due to the exercise. (You do not lose weight, you will probably gain some weight!)
- If you want to lose weight for instance, you need to avoid eating a lot of carbohydrates before working out. Your body will first use up your sugar stores before turning to “burning fat” for your energy production. This usually happens within 20-30 minutes of exercise. You will lose fat mass AND increase muscle mass due to the exercise.
Fatty acid facts!
I did a lot of thinking for this part – I didn’t want it to be too chemical, but I just couldn’t get around it for some parts! I ask you to bear with me, I will not give you an unnecessary chemistry lecture – I will however give you enough to understand what happens in your body!
Fatty acids play many roles, the most important 3 being:
- Fatty acids are an energy reservoir. They can broken down (or oxidized) to deliver that energy when required.
- Fatty acids are important building blocks for every cell in our body. (See article here)
- Fatty acids also serve as messengers, transporting signals throughout our body and prompting a specific response.
And how they affect each and every cell in our body can be found within their shape. Just stick with me for a page or two! I’ll keep it as unchemical as possible😊
Try and imagine the following chemical structures I explain as special legos! Use the individual lego pieces (atoms) to build a larger construct (molecules). (When I started thinking like that, I finally started to understand chemistry)
Fatty acids are long chains of carbon atoms that are linked one to another (Like beads on a string). Each carbon atom can form 4 bonds, meaning it can be attached to 4 different things.

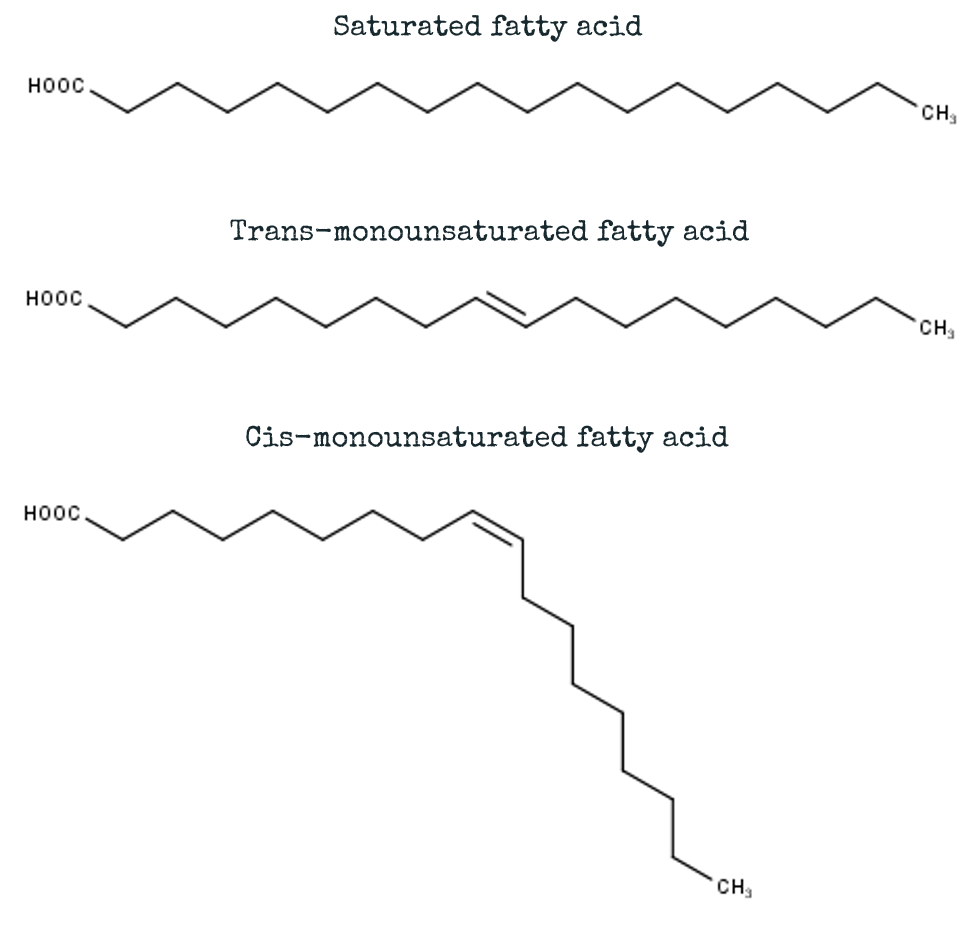
In a fatty acid chain, this would mean it is bound to the carbon before it and the carbon after it. The two remaining positions are filled with hydrogen! This makes it a “SATURATED” fatty acid, as it is saturated with hydrogen! Like a chain, a saturated fatty acid is linear in structure, and can be represented by a straight line (or a straight zigzag line, which is chemically more correct!). Saturated fatty acids are often found in animal fats like butter for example!
Sometimes, not all positions are filled with hydrogen atoms. That position, where the hydrogen is supposed to be, is empty and the bond tucks itself back between the carbon atoms. This is called a double bond! And fatty acids that contain such a double bond are called “UNSATURATED” fatty acids. “Unsaturated” because they do not contain the maximum amount of hydrogen atoms they could!
Now I am sure you have heard that unsaturated fatty acids are good for you in your diet – this is not completely true. I am sure you have heard about trans fatty acids, and that they are bad for you! Trans fatty acids or more commonly known as trans fats are also unsaturated fatty acids, however their shape is what separates them from the pack.
Let’s go back and have a look at that carbon chain, that is not filled with hydrogen. Instead of 4 hydrogen atoms on 2 carbons, they are now just two! If the remaining hydrogens are on opposite sides of the double bond (like shown in red in the figure above), then the double bond is in a “TRANS” configuration (Fig. B). These trans fatty acids don’t really occur naturally (or just in minimal amounts), they are rather produced by a process called hydrogenation – e.g. when healthy vegetable oils are synthetically hydrogenated to make them solid like with margarine.
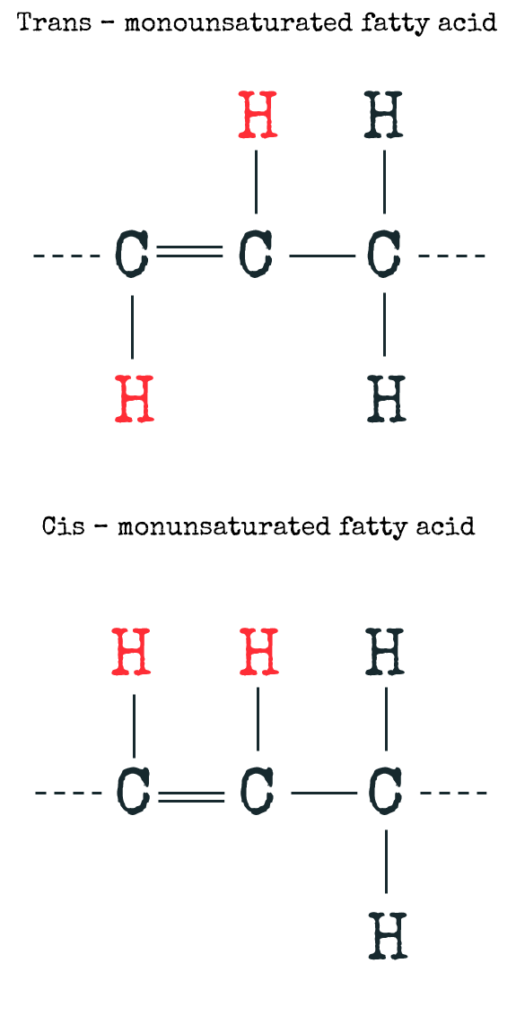
On the other hand, if the hydrogens are on the same side, it is in a “CIS” configuration (Fig. C)! Fatty acids in this cis configuration are usually referred to as unsaturated fatty acids when it comes to nutritional information. These types of unsaturated fatty acids are often found in vegetable oils like olive oil. These are the “healthy” ones! And this is what makes the difference in the structure and the shape!
SATURATED fatty acids as well as TRANS fatty acids are linear, whereas CIS fatty acids have a kink!
There’s more…essential fatty acids!
Hah! I actually got that question in my Master’s defense and I had a total blank (I did pass though, I promise:)! Well it did have its good side: I will never ever forget what an essential fatty acid is😊
Our body can produce a variety of fatty acids. However, some fatty acids that are required for good health cannot be produced by the body and need to be supplied by our diet. Those fatty acids are called “ESSENTIAL” fatty acids. The most common essential fatty acids are polyunsaturated ω-3 and ω-6 fatty acids (omega-3 and omega-6). They are called polyunsaturated fatty acids, as they contain more than one of those double bonds, that I explained earlier. They can be found in oily fish like makrel or salmon for example!
And there we go – another buzz-word used far too often in relation to our diet: omega-3 and omega-6 fatty acids! But what is it that makes these fatty acids so important? It is their structure!
Omega fatty acids contain more than 1 double bond, and are therefore called POLYunsaturated fatty acids. These multiple double bonds strongly affect the physical shape of the fatty acid and therefore also its properties.
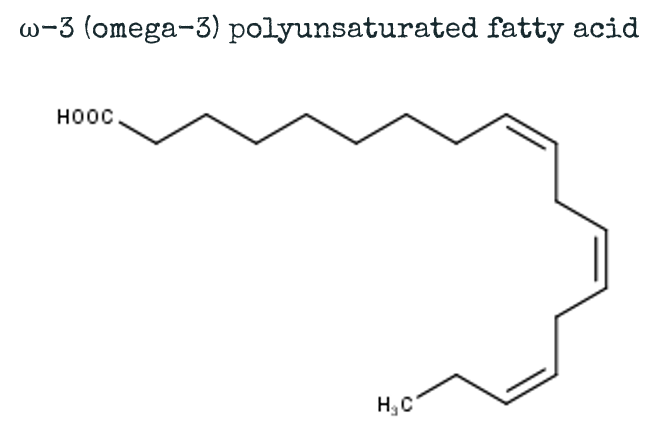
Further info: “Omega” refers to the end of the fatty acid carbon chain – an omega-3 fatty acid will have its first double bond on the third last carbon of the chain. An omega-6 fatty acid will have its first double bond on the 6th last carbon of the fatty acid etc…
Fatty acids are rarely found in their free form in the human body. They are usually bound as part of triglyceride molecules (energy storage) or phospholipids (structural components of cell membranes). For those of you who want to know more about how fatty acids are stored and metabolized, check out my article about fats in your body or the article about the secrets of a mediterranean diet! Or download the eBook to have all the information in one place!
I know not everybody has time to pop by the blog every day! But if you want to read the new recipes and articles then just follow Milly’s Melting Pot on Facebook, Instagram or Pinterest! Or register for the newsletter and get everything sent right to your inbox!
And if you are interested in how fats work in your body then check out this article in my blog section “The bubbly biochemist” !
(Bibliographie upon request)




